Online Database of Chemicals from Around the World
| Capot Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.capotchem.com | |||
 | +86 (571) 8558-6718 +86 13336195806 | |||
 | +86 (571) 8586-4795 | |||
 | capotchem@gmail.com sales@capotchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2006 | ||||
| Taizhou Crene Biotechnology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.pharm-intermediates.com | |||
 | +86 (576) 8881-3233 8820-5808 +86 13396860566 | |||
 | +86 (576) 8822-9589 | |||
 | sales@pharm-intermediates.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2009 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Zhejiang Warrant Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.zhejiang-warrant.com | |||
 | +86 (512) 8518-0611 +86 17312581805 | |||
 | +86 (512) 8917-1181 | |||
 | sales@zhejiang-warrant.com | |||
 | QQ Chat | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2011 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Amadis Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.amadischem.com | |||
 | +86 (571) 8992-5085 | |||
 | +86 (571) 8992-5065 | |||
 | sales@amadischem.com | |||
| Chemical manufacturer since 2010 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Linyi Lab Pharmaceutical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.labspharm.com | |||
 | +86 (539) 782-9635 | |||
 | +86 (539) 782-9635 | |||
 | Alex001@labspharm.com | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Coresyn Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.coresyn.com | |||
 | +86 (571) 8662-6709 | |||
 | sales@coresyn.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2016 | ||||
| Cangzhou Enke Pharma-tech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.enkepharma.com | |||
 | +86 (317) 510-5699 510-6597 +86 15533709196 | |||
 | +86 (317) 510-6596 | |||
 | sale@enkepharma.com enkepharma@126.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
 | WeChat: ymzhao | |||
| Chemical manufacturer since 2011 | ||||
| chemBlink Standard supplier since 2016 | ||||
| Shanghai Yingrui Biopharm Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.shyrchem.com | |||
 | +86 (21) 3358-5366 3466-6753 +86 13311639313 | |||
 | +86 (21) 3497-9012 | |||
 | sales02@shyrchem.com | |||
 | QQ Chat | |||
 | Skype Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Tianjin Reliable Pharmatech Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.reliablepharmatech.com | |||
 | +86 (22) 2856-7687 | |||
 | sales@reliablepharmatech.com | |||
| Chemical distributor since 2017 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Shanghai Fuxin Pharmaceutical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fuxinpharm.com | |||
 | +86 (21) 3130-0828 +86 18645121291 | |||
 | +86 (21) 3130-0828 | |||
 | contact@fuxinpharm.com | |||
| Chemical manufacturer since 2016 | ||||
| chemBlink Standard supplier since 2018 | ||||
| Tecoland Corporation | USA | |||
|---|---|---|---|---|
 | www.tecoland.com | |||
 | +1 (732) 603-9577 | |||
 | +1 (732) 906-1522 | |||
 | info@tecoland.com | |||
| Chemical distributor since 2001 | ||||
| chemBlink Standard supplier since 2019 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | API >> Synthetic anti-infective drugs >> Antifungal drugs |
|---|---|
| Name | Tedizolid phosphate |
| Synonyms | TR 701FA; (5R)-3-[3-Fluoro-4-[6-(2-methyl-2H-tetrazol-5-yl)-3-pyridinyl]phenyl]-5-[(phosphonooxy)methyl]-2-oxazolidinone |
| Molecular Structure | 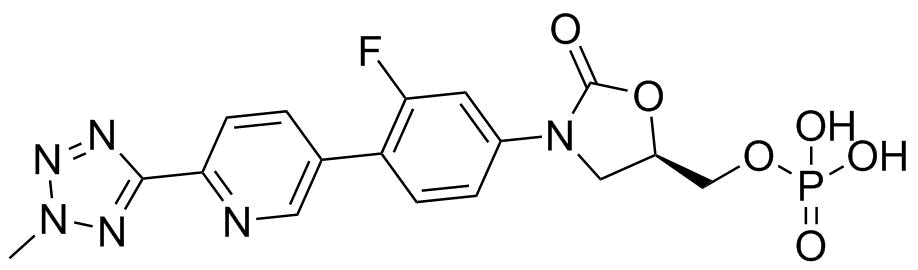 |
| Molecular Formula | C17H16FN6O6P |
| Molecular Weight | 450.32 |
| CAS Registry Number | 856867-55-5 |
| EC Number | 836-194-6 |
| SMILES | CN1N=C(N=N1)C2=NC=C(C=C2)C3=C(C=C(C=C3)N4C[C@@H](OC4=O)COP(=O)(O)O)F |
| Solubility | Very slightly soluble (0.68 g/L) (25 $degree$C), Calc.* |
|---|---|
| Density | 1.75$+/-$0.1 g/cm3 (20 $degree$C 760 Torr), Calc.* |
| Boiling point | 725.6$+/-$70.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 392.6$+/-$35.7 $degree$C (Calc.)* |
| Index of refraction | 1.739 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS08;GHS09 Warning Details GHS08;GHS09 Warning Details | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H361-H373-H400-H410 Details | ||||||||||||||||||||
| Safety Statements | P203-P260-P273-P280-P318-P319-P391-P405-P501 Details | ||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||
| |||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||
|
Tedizolid phosphate is the phosphate ester prodrug of tedizolid, an oxazolidinone-class antibacterial agent used for the treatment of acute bacterial skin and skin structure infections. It is administered orally or intravenously and is rapidly converted in vivo to the active form, tedizolid. Structurally, tedizolid phosphate contains an oxazolidinone core, which is characteristic of this antibiotic class. The oxazolidinone ring is a five-membered heterocycle containing both nitrogen and oxygen atoms, and it plays a central role in antibacterial activity. The phosphate group in tedizolid phosphate is attached as a hydrophilic moiety that enhances aqueous solubility, improving formulation suitability for intravenous administration. Once administered, endogenous phosphatases cleave the phosphate ester, releasing the active drug tedizolid. Tedizolid exerts its antibacterial effect by inhibiting bacterial protein synthesis. It binds to the 50S subunit of the bacterial ribosome, specifically near the peptidyl transferase center, preventing the formation of the initiation complex required for protein translation. This mechanism blocks the early stages of protein synthesis, leading to inhibition of bacterial growth. Because this binding site is distinct from many other antibiotic classes, oxazolidinones are generally effective against strains resistant to other protein synthesis inhibitors. Tedizolid phosphate was developed as an improvement over earlier oxazolidinones such as linezolid. Structural modifications to the core molecule were designed to enhance potency, extend the duration of action, and reduce certain side effects associated with earlier drugs in the class. These modifications include optimized aromatic substituents and heterocyclic features that improve binding affinity to the ribosomal target. The phosphate prodrug form was specifically introduced to improve pharmacokinetic properties. Tedizolid itself has limited water solubility, whereas the phosphate ester is significantly more soluble, allowing for easier formulation in injectable solutions. After administration, the prodrug is rapidly dephosphorylated by alkaline phosphatases present in blood and tissues, yielding the active antimicrobial compound. Clinically, tedizolid phosphate is used primarily for short-course treatment of acute bacterial skin and skin structure infections caused by susceptible Gram-positive organisms, including Staphylococcus aureus and Streptococcus species. It is active against methicillin-resistant Staphylococcus aureus (MRSA), making it useful in settings where resistant infections are a concern. The mechanism of action of tedizolid involves inhibition of protein synthesis at an early stage, which reduces the likelihood of cross-resistance with other antibiotic classes such as macrolides, beta-lactams, or fluoroquinolones. Its binding site on the ribosome overlaps with but is not identical to that of other oxazolidinones, contributing to its antibacterial profile. From a pharmacokinetic perspective, tedizolid phosphate is rapidly converted to tedizolid, which has good oral bioavailability and tissue penetration. The active drug exhibits a long half-life, allowing for once-daily dosing and short treatment courses in clinical use. Adverse effects associated with tedizolid are generally similar to those of other oxazolidinones but may occur with lower frequency depending on treatment duration. These can include gastrointestinal symptoms, headache, and potential hematologic effects such as thrombocytopenia with prolonged use. However, short-course therapy is designed to minimize such risks. Overall, tedizolid phosphate is a water-soluble prodrug of the oxazolidinone antibiotic tedizolid. It functions as a ribosomal protein synthesis inhibitor targeting Gram-positive bacteria and is clinically used for the treatment of acute skin and soft tissue infections. Its prodrug design and structural optimization reflect advances in antibiotic development aimed at improving potency, safety, and dosing convenience. References 2025. Tedizolid for osteoarticular infections: Evaluation of the published evidence. European Journal of Pharmacology. URL: https://pubmed.ncbi.nlm.nih.gov/40090537 2025. A Phase 3 Study of the Safety and Efficacy of Tedizolid Phosphate in Patients <12 Years of Age With Acute Bacterial Skin and Skin Structure Infections. The Pediatric infectious disease journal. URL: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC12058361 2024. In vitro and in vivo evaluation of tedizolid nanoparticle incorporated buccal films for oromucosal infections. International Journal of Pharmaceutics. DOI: 10.1016/j.ijpharm.2024.124688 |
| Market Analysis Reports |