Online Database of Chemicals from Around the World
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Nantong C&R Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.chenrunchem.com | |||
 | +86 (513) 8874-3581 +86 15358700783 | |||
 | +86 (513) 8297-1198 | |||
 | chenrunchem@sina.com 909600256@qq.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2009 | ||||
| chemBlink Standard supplier since 2013 | ||||
| Guangzhou Jun Xin Chemical Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.jxinchem.com | |||
 | +86 (020) 8256-9208 | |||
 | +86 (020) 2826-9100 | |||
 | ivyyoung@gzjxchem.com | |||
 | QQ Chat | |||
 | WeChat: gzjx18122041046 | |||
 | WhatsApp:+8618122041046 | |||
| Chemical distributor since 2007 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Crescent Chemical Co. Inc. | USA | |||
|---|---|---|---|---|
 | www.crescentchemical.com | |||
 | +1 (631) 348-0333 | |||
 | +1 (631) 348-0913 | |||
 | crescent@creschem.com | |||
| Chemical distributor | ||||
| Classification | Chemical reagent >> Organic reagent >> Ether |
|---|---|
| Name | Ethoxylated isodecanol |
| Synonyms | Caflon DE 0600; Chemal DA 6; Chemal DA 9; Correctol SE; Emulphogene DA 530; Ethoxylated isodecanol; Ethoxylated isodecyl alcohol; Ethylan CD 109; FC 2; Finesurf D 45; Finesurf D 60; Finesurf D 65; Finesurf D 85; Genapol ID 60; ID 206; Iconol DA 6; Igepal DA 530; LT 601; Lutensol XP 89; Lutensol XP 99; Marlipal 013Ei; Mogiol; Naroacty ID 60; Nissan Nonion ID 206; Noigen SD 110; Noigen SD 150; Noigen SD 30; Noigen SD 300; Noigen SD 60; Noigen SD 80; Noigen SDX 60; Noigen XL 1000F; Noigen XL 140; Noigen XL 400; Nonion ID 206; Polyethylene glycol isodecyl ether; Polyethylene glycol isodecyl monoether; Polyethylene glycol monoether with Exxal 10; Polyethylene glycol monoisodecyl ether; Polyoxyethylene monoisodecyl ether; Rhodasurf 860P; Rhodasurf DA 530; Rhodasurf DA 630; Rhodasurf DA 630E; Rhodasurf DA 639; SD 110; SDX 60; Safetycut ID 1033; Safetycut ID 1061; Serdox NDL 9.8; Simulsol NW 900; Stepantex DA 6; Surfonic DA 6; Synperonic 10/11; Synperonic 10/5; Synperonic 10/7; Trend 90; Trend 90EC; Trycol 5952; Trycol LF 1; Walloxen ID 110; XP 70 |
| Molecular Structure | 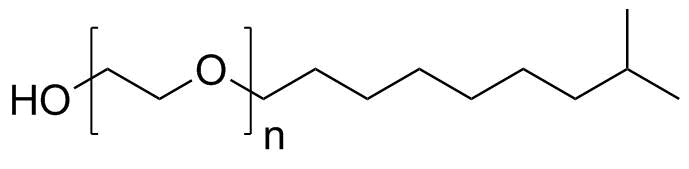 |
| Molecular Formula | (C2H4O)n.C10H22O |
| CAS Registry Number | 61827-42-7 |
| EC Number | 612-519-5 |
| SMILES | CC(C)CCCCCCCOCCO |
| Density | 0.9$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 268.0$+/-$8.0 $degree$C 760 mmHg (Calc.)* |
| Flash point | 72.4$+/-$7.7 $degree$C (Calc.)* |
| Index of refraction | 1.44 (Calc.)* |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |   GHS05;GHS07 Danger Details GHS05;GHS07 Danger Details | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H318 Details | ||||||||||||||||||||||||
| Safety Statements | P264-P264+P265-P270-P280-P301+P317-P305+P354+P338-P317-P330-P501 Details | ||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||
| |||||||||||||||||||||||||
|
Ethoxylated isodecanol is a nonionic surfactant derived from isodecanol, a branched C10 alcohol, that has been reacted with ethylene oxide to introduce hydrophilic polyoxyethylene (–CH2CH2O–)n chains. The resulting structure combines a hydrophobic branched alkyl group with a hydrophilic ethoxylate chain, giving the molecule amphiphilic properties that are widely used in industrial and formulation chemistry. The base molecule, isodecanol, is a mixture of branched-chain decyl alcohol isomers produced through hydroformylation and hydrogenation processes from olefin precursors. Its branched structure provides improved fluidity and lower crystallization tendency compared with linear alcohols. Ethoxylation is carried out by reacting isodecanol with ethylene oxide under alkaline or catalytic conditions, producing a distribution of ethoxylated homologues with varying numbers of ethylene oxide units. The degree of ethoxylation (n) determines the hydrophilic–lipophilic balance (HLB) and thus the surfactant’s functional properties. The molecular structure consists of a hydrophobic isodecyl group (C10 branched alkyl chain) attached through an ether linkage to a polyoxyethylene chain. The ethoxylate segment is highly polar due to multiple ether oxygen atoms capable of hydrogen bonding with water. This combination allows the molecule to localize at interfaces between oil and water phases, reducing interfacial tension and stabilizing emulsions. Ethoxylated isodecanol belongs to the class of nonionic surfactants, which do not carry a formal charge in solution. This distinguishes it from anionic or cationic surfactants and makes it less sensitive to changes in pH and electrolyte concentration. As a result, it is widely used in formulations that require stability across varying chemical environments. In aqueous systems, ethoxylated isodecanol molecules can form micelles above their critical micelle concentration. In these aggregates, the hydrophobic isodecyl groups cluster inward, while the ethoxylated chains interact with water. This property enables solubilization of hydrophobic substances such as oils, fragrances, and organic contaminants. The compound is widely used in industrial, household, and institutional cleaning products as an emulsifier, wetting agent, and detergent component. It is also used in textile processing, agricultural formulations, and coatings, where it helps disperse hydrophobic ingredients and improve surface wetting. Its branched hydrophobic structure provides good low-temperature performance and resistance to crystallization, which is advantageous in liquid formulations. In comparison with linear alcohol ethoxylates, ethoxylated isodecanol often exhibits improved biodegradation profiles and better low-foam characteristics depending on ethoxylation degree. These properties make it suitable for applications where controlled foaming or rapid rinsing is required. The physicochemical behavior of ethoxylated isodecanol is strongly influenced by the balance between the hydrophobic alkyl chain and the hydrophilic ethoxylate segment. Increasing the number of ethylene oxide units increases water solubility and hydrophilicity, while decreasing it enhances oil solubility and interfacial activity. Overall, ethoxylated isodecanol is a versatile nonionic surfactant produced by ethoxylation of branched isodecanol. Its amphiphilic structure provides effective emulsifying, wetting, and solubilizing properties, making it widely used in industrial formulations, cleaning systems, and specialty chemical applications. References 2016. GST activity and membrane lipid saturation prevents mesotrione-induced cellular damage in Pantoea ananatis. AMB Express. DOI: 10.1186/s13568-016-0240-x |
| Market Analysis Reports |