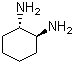
(1S,2S)-(+)-1,2-Diaminocyclohexane is a stereochemically defined vicinal diamine with the molecular formula C6H14N2. It consists of a cyclohexane ring bearing two amino groups attached to adjacent carbon atoms in the 1 and 2 positions. The compound is one of the optically active stereoisomers of 1,2-diaminocyclohexane and possesses the absolute configuration (1S,2S), giving rise to chirality and optical rotation.
The cyclohexane ring typically adopts a chair conformation, which is the most stable conformation for saturated six-membered rings. In the (1S,2S) isomer, the stereochemical arrangement of the amino groups creates a defined three-dimensional geometry that strongly influences the molecule’s coordination behavior and reactivity. Because both carbon atoms bearing amino substituents are stereogenic centers, the compound exists as a specific enantiomer rather than a racemic mixture.
The compound belongs to the class of vicinal diamines, molecules containing two amino groups on neighboring carbon atoms. The amino groups are strongly basic and nucleophilic, allowing the molecule to participate in a wide range of reactions including protonation, condensation, alkylation, acylation, and coordination to metal ions.
One of the most important properties of (1S,2S)-(+)-1,2-diaminocyclohexane is its role as a chiral ligand in coordination chemistry and asymmetric synthesis. The rigid cyclohexane framework fixes the spatial orientation of the two nitrogen donor atoms, enabling formation of stereochemically defined metal complexes. These complexes can induce asymmetry in catalytic reactions, making the compound valuable in enantioselective catalysis.
The diamine can coordinate to transition metals through both nitrogen atoms, forming stable five-membered chelate rings. The chirality of the ligand is transferred to the metal coordination sphere, which can influence the stereochemical outcome of catalytic transformations. As a result, derivatives of (1S,2S)-1,2-diaminocyclohexane have been widely used in asymmetric hydrogenation, epoxidation, and carbon–carbon bond-forming reactions.
The compound has also served as an important building block in the synthesis of chiral auxiliaries, pharmaceuticals, and specialty ligands. The defined stereochemistry and conformational rigidity of the cyclohexane ring make it useful for controlling molecular geometry in stereoselective synthesis.
Industrial and laboratory synthesis of enantiomerically enriched (1S,2S)-(+)-1,2-diaminocyclohexane may involve resolution of racemic mixtures or stereoselective synthesis from chiral precursors. Catalytic hydrogenation of appropriately substituted aromatic or unsaturated intermediates has also been employed to establish the required stereochemistry.
From a physicochemical perspective, the compound is highly polar because of the two primary amino groups. It readily forms salts with mineral and organic acids and participates extensively in hydrogen bonding. The molecule is generally soluble in water and polar organic solvents.
The stereochemical designation “(+)" indicates that the compound rotates plane-polarized light in the positive direction under specified measurement conditions. Optical activity is an important feature in applications involving asymmetric catalysis and chiral recognition.
Historically, chiral diamines such as (1S,2S)-(+)-1,2-diaminocyclohexane became increasingly important with the development of asymmetric synthesis in the late 20th century. Their ability to induce chirality in catalytic systems contributed significantly to advances in enantioselective organic chemistry and homogeneous catalysis.
Overall, (1S,2S)-(+)-1,2-diaminocyclohexane is a chiral vicinal diamine characterized by a rigid cyclohexane framework and two stereochemically defined amino substituents. Its strong metal-binding ability and well-defined three-dimensional structure make it an important ligand and intermediate in coordination chemistry, asymmetric catalysis, and stereoselective synthesis.
References
2026. Helical wrapping and charge-transfer driven multi-stranded crystalline helices from a twisted figure-of-eight macrocycle. Science China Materials.
DOI: 10.1007/s40843-025-3830-6
2023. Non-conjugated Polynorbornene Hosts with High Triplet Energy Levels for Solution-processed Narrowband Blue OLEDs. Chemical Research in Chinese Universities.
DOI: 10.1007/s40242-023-3167-1
2022. An Improved Scalable Process for the Synthesis of (S,S)-DACH-Ph Trost Ligand. Synlett.
DOI: 10.1055/s-0041-1738371
|






























































 GHS05;GHS07 Danger Details
GHS05;GHS07 Danger Details