Online Database of Chemicals from Around the World
| F&F Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.fnfchem.com | |||
 | +86 (10) 6444-6910 | |||
 | +86 (10) 6444-6915 | |||
 | heyifnfchem@163.com heyi@fnfchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1997 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Mascot I.E. CO.,Ltd. | China | |||
|---|---|---|---|---|
 | www.mascotchem.com | |||
 | +86 (519) 8501-0339 +86 13584504415 | |||
 | +86 (519) 8501-0366 | |||
 | info@mascotchem.com | |||
 | QQ Chat | |||
| Chemical distributor since 2006 | ||||
| chemBlink Standard supplier since 2006 | ||||
| Simagchem Corporation | China | |||
|---|---|---|---|---|
 | www.simagchem.com | |||
 | +86 13806087780 | |||
 | +86 (592) 268-0237 | |||
 | sale@simagchem.com | |||
| Chemical manufacturer since 2002 | ||||
| chemBlink Standard supplier since 2008 | ||||
| Hefei TNJ Chemical Industry Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.tnjchem.com | |||
 | +86 (551) 6541-8684 | |||
 | +86 (551) 6541-8697 | |||
 | sales@tnjchem.com | |||
| Chemical manufacturer since 2001 | ||||
| chemBlink Standard supplier since 2010 | ||||
| BOC Sciences | USA | |||
|---|---|---|---|---|
 | www.bocsci.com | |||
 | +1 (631) 485-4226 | |||
 | +1 (631) 614-7828 | |||
 | info@bocsci.com | |||
| Chemical manufacturer | ||||
| chemBlink Standard supplier since 2010 | ||||
| Hangzhou Leap Chem Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.leapchem.com | |||
 | +86 (571) 8771-1850 | |||
 | market19@leapchem.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 2006 | ||||
| chemBlink Standard supplier since 2015 | ||||
| Hangzhou Hairui Chemical Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.hairuichem.com | |||
 | +86 (571) 8669-1155 | |||
 | +86 (571) 8669-1154 | |||
 | sales@hairuichem.com | |||
| Chemical distributor since 2005 | ||||
| chemBlink Standard supplier since 2017 | ||||
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Scientific Polymer Products, Inc. | USA | |||
|---|---|---|---|---|
 | www.scientificpolymer.com | |||
 | +1 (585) 265-0413 | |||
 | +1 (585) 265-1390 | |||
 | custserv@scipoly.com | |||
| Chemical manufacturer | ||||
| Shandong Xingshun New Material Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.sd-xingshun.com | |||
 | +86 (519) 8646-1196 +86 (519) 8646-4994 +86 13809078067 | |||
 | +86 (519) 8646-3703 | |||
 | gxx@xingshengtech.com gxy@xingshengtech.com xp@xingshengtech.com | |||
 | QQ Chat | |||
| Chemical manufacturer since 1996 | ||||
| Organica Feinchemie GmbH | Germany | |||
|---|---|---|---|---|
 | www.organica.de | |||
 | +49 (3494) 63-6215 | |||
 | +49 (3494) 63-6165 | |||
 | dmi@weylchem-organica.com | |||
| Chemical manufacturer | ||||
| Classification | Organic raw materials >> Hydrocarbon compounds and their derivatives >> Aromatic hydrocarbon |
|---|---|
| Name | 4-Vinylbenzyl chloride |
| Synonyms | 4-Chloromethyl styrene |
| Molecular Structure | 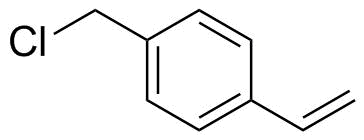 |
| Molecular Formula | C9H9Cl |
| Molecular Weight | 152.62 |
| CAS Registry Number | 1592-20-7 |
| EC Number | 216-471-2 |
| SMILES | C=CC1=CC=C(C=C1)CCl |
| Density | 1.1$+/-$0.1 g/cm3 Calc.* |
|---|---|
| Boiling point | 228.9$+/-$9.0 $degree$C 760 mmHg (Calc.)*, 229 $degree$C (Expl.) |
| Flash point | 105.0 $degree$C (Calc.)* |
| Index of refraction | 1.57 (Calc.)*, 1.572 (Expl.) |
| * | Calculated using Advanced Chemistry Development (ACD/Labs) Software. |
| Hazard Symbols |    GHS05;GHS06;GHS07 Danger Details GHS05;GHS06;GHS07 Danger Details | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302-H311-H312-H314-H317 Details | ||||||||||||||||||||||||||||||||||||||||
| Safety Statements | P260-P261-P262-P264-P270-P272-P280-P301+P317-P301+P330+P331-P302+P352-P302+P361+P354-P304+P340-P305+P354+P338-P316-P317-P321-P330-P333+P317-P361+P364-P362+P364-P363-P405-P501 Details | ||||||||||||||||||||||||||||||||||||||||
| Hazard Classification | |||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||
| Transport Information | UN 3265 | ||||||||||||||||||||||||||||||||||||||||
| SDS | Available | ||||||||||||||||||||||||||||||||||||||||
|
4-Vinylbenzyl chloride is an aromatic organic compound consisting of a benzene ring bearing two substituents in the para position: a vinyl group (–CH=CH2) and a benzylic chloromethyl group (–CH2Cl). Its molecular structure combines two distinct reactive functionalities, making it a versatile monomer and intermediate in polymer and organic synthesis. The compound belongs to the family of vinylbenzyl halides, which are bifunctional aromatic derivatives widely used in polymer chemistry. The vinyl group is capable of undergoing free-radical polymerization, while the benzylic chloride group can participate in nucleophilic substitution reactions. This dual reactivity allows 4-vinylbenzyl chloride to be incorporated into polymer backbones while retaining a chemically active side chain for further functionalization. Structurally, the benzene ring provides an aromatic scaffold that stabilizes the substituents through conjugation and inductive effects. The vinyl group is conjugated with the aromatic ring to some extent, which influences its reactivity in polymerization processes. The chloromethyl group, attached to the benzylic position, is activated toward nucleophilic substitution due to the resonance stabilization of the benzylic intermediate formed during reaction. The development and use of vinylbenzyl chloride derivatives emerged alongside advances in polymer chemistry in the mid-20th century. Researchers sought monomers that could form stable polymer backbones while also allowing post-polymerization modification. 4-Vinylbenzyl chloride became particularly valuable because it could be polymerized into polystyrene-like materials while introducing reactive chloromethyl groups into the resulting polymer. Synthesis of 4-vinylbenzyl chloride typically involves preparation of a para-substituted benzyl chloride precursor followed by introduction of the vinyl group. One common route includes chloromethylation of p-vinyltoluene or related aromatic precursors, although care must be taken to prevent polymerization of the vinyl group during synthesis. Industrial processes are designed to control these competing reactivities and achieve selective formation of the desired monomer. In polymer chemistry, 4-vinylbenzyl chloride is widely used as a functional monomer in copolymerization reactions. It is often copolymerized with styrene, acrylates, or other vinyl monomers using free-radical initiators. The resulting polymers contain pendant chloromethyl groups along the backbone, which can be chemically modified after polymer formation. One of the most important applications of polymers derived from 4-vinylbenzyl chloride is in the production of ion-exchange resins. The chloromethyl group can be reacted with tertiary amines to form quaternary ammonium groups, yielding strongly basic anion-exchange materials. These resins are widely used in water purification, chemical separation, and industrial catalysis. In addition to ion-exchange applications, polymers containing vinylbenzyl chloride units are used as intermediates for crosslinked polymer networks. The benzylic chloride functionality allows further chemical modification, enabling the introduction of a wide range of functional groups such as alcohols, amines, or carboxylic acids. This versatility makes the compound useful in designing tailored polymer materials. The vinyl group also allows for controlled radical polymerization, enabling incorporation into well-defined polymer architectures. This has been important in the development of advanced materials such as functional coatings, membrane materials, and support matrices for catalysts. Overall, 4-vinylbenzyl chloride is a bifunctional aromatic monomer that combines a polymerizable vinyl group with a reactive benzylic chloride substituent. Its development has played a significant role in functional polymer chemistry, particularly in the synthesis of ion-exchange resins and chemically modifiable polymer materials. Its dual reactivity continues to make it a valuable building block in materials science and organic synthesis. References 2026. Cross-linker strategies for high-performance AEMs: balancing conductivity, stability, and fuel cell efficiency. Chemical Papers. DOI: 10.1007/s11696-026-04695-8 2025. Chemoselective Reduction of Aryl Halide Bearing Nitro Group Enabled by Heterogeneous Monolith Supported Pd Catalyst. Topics in Catalysis. DOI: 10.1007/s11244-025-02242-5 2025. Synthesis and characterization of poly(4-vinylbenzyl-graft-ethylene glycol methyl ether) amphiphilic graft copolymer via free-radical polymerization and ‘click’ chemistry. Journal of Chemical Sciences. DOI: 10.1007/s12039-025-02368-9 |
| Market Analysis Reports |