Online Database of Chemicals from Around the World
| Ningbo Wingoo Semiconductor Core Material Technology Co., Ltd. | China | |||
|---|---|---|---|---|
 | www.wingooxc.com | |||
 | +86 18842662104 | |||
 | zoulin@wingooxc.com | |||
| Chemical manufacturer since 2025 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Classification | Chemical reagent >> Organic reagent >> Azo, diazonium compound |
|---|---|
| Name | Bis(cyclohexylsulfonyl)diazomethane |
| Synonyms | [cyclohexylsulfonyl(diazo)methyl]sulfonylcyclohexane |
| Molecular Structure | 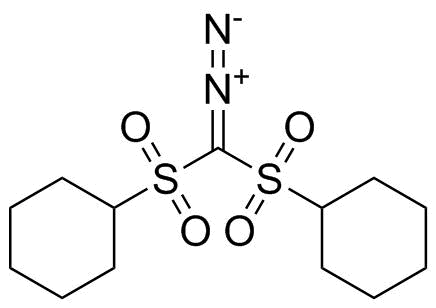 |
| Molecular Formula | C13H22N2O4S2 |
| Molecular Weight | 334.45 |
| CAS Registry Number | 138529-81-4 |
| EC Number | 689-810-9 |
| SMILES | C1CCC(CC1)S(=O)(=O)C(=[N+]=[N-])S(=O)(=O)C2CCCCC2 |
| Hazard Classification | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| |||||||||
|
Bis(cyclohexylsulfonyl)diazomethane is a sulfone-stabilized diazo compound in which a central diazomethyl group (–CHN2) is flanked by two cyclohexylsulfonyl (–SO2–C6H11) substituents. This arrangement places the diazo functionality between two strongly electron-withdrawing sulfonyl groups, which significantly stabilizes the otherwise highly reactive diazo moiety and modulates its reactivity in organic synthesis. The molecular structure features a central carbon atom bonded to a diazo group (–N≡N) and two sulfonyl-substituted cyclohexyl groups. The sulfonyl groups withdraw electron density, stabilizing negative charge development and enabling controlled generation of reactive intermediates such as carbenes or carbanions under appropriate conditions. The cyclohexyl rings provide steric bulk, which can influence selectivity in reactions and improve handling stability compared to simpler diazo compounds. Synthesis of bis(cyclohexylsulfonyl)diazomethane typically involves preparation of the corresponding bis(sulfonyl)methane precursor followed by diazo transfer or oxidation to introduce the diazo functionality. Reaction conditions are carefully controlled due to the inherent reactivity of diazo compounds, and purification is performed to obtain a stable, crystalline product suitable for use in synthesis. This compound is primarily used as a reagent in organic chemistry. It can act as a precursor to metal carbenes in the presence of transition-metal catalysts, enabling transformations such as cyclopropanation of alkenes, insertion into C–H or heteroatom–H bonds, and ylide formation. The electron-withdrawing sulfonyl groups help regulate carbene reactivity, providing improved selectivity compared to less stabilized diazo reagents. In addition, bis(cyclohexylsulfonyl)diazomethane can participate in nucleophilic substitution and condensation reactions, serving as a synthetic equivalent for methylene transfer under controlled conditions. Its stability relative to simpler diazomethane makes it safer to handle, though it still requires careful control due to potential decomposition and gas evolution. Overall, bis(cyclohexylsulfonyl)diazomethane is a stabilized diazo reagent that combines the reactivity of diazo chemistry with enhanced control provided by electron-withdrawing sulfonyl groups. Its ability to generate carbenes and participate in selective transformations makes it a valuable tool in modern organic synthesis. References 2023. Development of heat-responsive adhesive materials that are stable during use and quickly deteriorate during dismantling. Polymer Journal. DOI: 10.1038/s41428-023-00849-7 |
| Market Analysis Reports |