Online Database of Chemicals from Around the World
| Cfm Oskar Tropitzsch GmbH | Germany | |||
|---|---|---|---|---|
 | www.cfmot.de | |||
 | +49 (9231) 9619-0 | |||
 | +49 (9231) 9619-60 | |||
 | info@cfmot.de | |||
| Chemical distributor since 1985 | ||||
| chemBlink Standard supplier since 2022 | ||||
| Pure Bio Medical Jiangsu Ltd. | China | |||
|---|---|---|---|---|
 | www.purejs.cn | |||
 | +86 (025) 8560-7383 | |||
 | jiangziwei@purejs.cn | |||
| Chemical manufacturer since 2019 | ||||
| chemBlink Standard supplier since 2026 | ||||
| Anvia Chemicals, LLC | USA | |||
|---|---|---|---|---|
 | www.anviachem.com | |||
 | +1 (414) 534-7845 | |||
 | +1 (414) 762-5539 | |||
 | sales@anviachem.com | |||
| Chemical manufacturer | ||||
| Acros Organics | Belgium | |||
|---|---|---|---|---|
 | www.acros.com | |||
 | +86 (21) 5258-1100 | |||
 | +86 (21) 5258-0119 | |||
 | info@acros.com | |||
| Chemical manufacturer | ||||
| Classification | Biochemical >> Biochemical reagent >> Acid-base indicator |
|---|---|
| Name | Methylene Blue |
| Synonyms | 3,7-Bis(dimethylamino)phenothiazin-5-ium chloride |
| Molecular Structure | 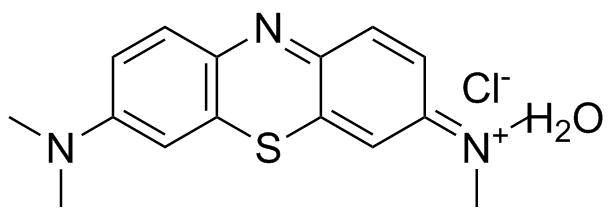 |
| Molecular Formula | C16H18ClN3S |
| Molecular Weight | 319.85 |
| CAS Registry Number | 122965-43-9 |
| EC Number | 602-909-3 |
| SMILES | CN(C)C1=CC2=C(C=C1)N=C3C=CC(=[N+](C)C)C=C3S2.O.[Cl-] |
| Melting point | 100-110 $degree$C |
|---|---|
| Hazard Symbols |  GHS07 Warning Details GHS07 Warning Details | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Risk Statements | H302 Details | ||||||||||||||||
| Safety Statements | P264-P270-P301+P317-P330-P501 Details | ||||||||||||||||
| Hazard Classification | |||||||||||||||||
| |||||||||||||||||
| SDS | Available | ||||||||||||||||
|
Methylene Blue is a heterocyclic aromatic chemical compound belonging to the phenothiazinium dye class. It is a cationic thiazine dye with the molecular formula C16H18N3SCl in its commonly used chloride salt form. The compound is characterized by a tricyclic phenothiazine core containing nitrogen and sulfur heteroatoms, with dimethylamino substituents that contribute to its strong coloration and redox activity. Methylene Blue was first synthesized in 1876 by Heinrich Caro at BASF during early industrial dye development. It was among the first synthetic dyes produced from coal tar derivatives and played a significant role in the emergence of modern dye chemistry. Initially developed for textile applications, it soon gained importance in biological and medical research due to its strong staining properties and redox behavior. The structure of Methylene Blue consists of a phenothiazine skeleton, where two benzene rings are connected through a sulfur and nitrogen-containing central ring system. The positively charged nitrogen atom gives the molecule its cationic nature, allowing it to bind strongly to negatively charged biological structures such as nucleic acids and acidic tissue components. This property is responsible for its effectiveness as a biological stain. In biological applications, Methylene Blue has been widely used as a histological dye for staining cells and tissues. It was one of the early dyes used in microscopy to visualize cellular structures, including nuclei and bacteria. Its ability to selectively bind to acidic components made it valuable in early microbiology and pathology. In addition to its staining properties, Methylene Blue exhibits redox activity. It can undergo reversible reduction to leucomethylene blue, a colorless form. This reversible redox behavior has been exploited in biochemical assays and analytical chemistry. The compound can act as an electron carrier in redox reactions, which has contributed to its use in studies of cellular respiration and metabolic processes. Methylene Blue has also been used in medicine for more than a century. It has been applied as a treatment for methemoglobinemia, a condition in which hemoglobin is oxidized to a form that cannot effectively transport oxygen. In this context, Methylene Blue acts as an electron donor, helping to restore hemoglobin to its functional state. It has also been used as a urinary antiseptic and in certain diagnostic procedures. In modern biomedical research, Methylene Blue has been studied for a variety of potential therapeutic effects, including its role in mitochondrial function, neuroprotection, and as a photosensitizer in antimicrobial photodynamic therapy. Its ability to interact with biological redox systems has made it a compound of continued scientific interest. In industrial contexts, Methylene Blue remains an important dye for analytical chemistry, including its use in titrations and adsorption studies. It is also used as a tracer compound in environmental and engineering studies due to its strong color and detectability at low concentrations. Overall, Methylene Blue is a historically significant phenothiazinium dye with broad applications spanning chemistry, biology, medicine, and industry. Its unique combination of structural features, strong coloration, and reversible redox behavior has made it one of the most widely studied and utilized synthetic dyes since its discovery in the 19th century. References 2025. CaTiO3/TiO2 Nanocomposite: Easy Synthesis, Structure, Optical and Photocatalytic Activity. Topics in Catalysis. DOI: 10.1007/s11244-025-02235-4 2025. Efficient Degradation of Methylene Blue by Heterogeneous Fenton Reaction Using FeWO4 Nanocatalysts. International Journal of Environmental Research. DOI: 10.1007/s41742-025-00905-5 2025. Cationic Dye Treatment Using Local Tunisian Iron Tailings for Synthetic and Textile Wastewater. Chemistry Africa. DOI: 10.1007/s42250-025-01354-5 |
| Market Analysis Reports |